 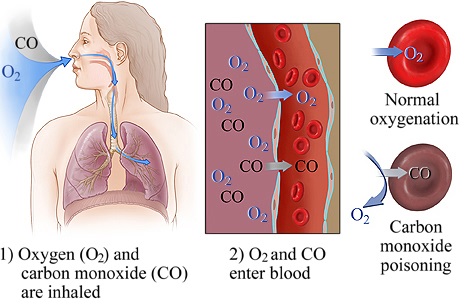 
HS has also been said to have this smell, whereas CN has been described to have a "bitter burning taste." 2,9 Many odorless simple asphyxiants such as propane are used as fuels and are injected with a substance with a "rotten egg scent" during manufacturing. For conscious patients, it can be helpful to ask if they recall any distinct odors prior to symptom onset. If they were brought in after a fire, found in a confined space, or found in any industrial-type setting, asphyxiant toxicity should be included in the differential. These patients may be found unconscious, and if brought in by ambulance, questions to ask EMS should include the circumstances in which the patient was found. First, a good history is essential in directing treatment. When encountering a patient who potentially suffered asphyxiant poisoning, several steps in management should be taken regardless of exposure. These are mostly related to industrial exposure while working in confined spaces. 1,6,7 Exposure to non-CO simple asphyxiants is less common, with the American Association of Poison Control Centers reporting about 2,500 cases in 2016. 4,5 HS is a byproduct formed by the decay of organic material and the exposure risk is usually occupational, particularly for those working in oil drilling or with manure. 3 CN has been associated with occupational exposure in mining and manufacturing, and up to 35% of fire-related inhalational injuries are linked to CN toxicity. The most common source of asphyxiant poisoning is CO, with more than 50,000 cases annually in the United States primarily related to fires, motor exhaust, and gas leak. 2 If untreated, asphyxiant exposure can lead to myocardial damage, permanent neurological deficits, and death. Unlike simple asphyxiants, systemic asphyxiants are not limited to gases, and include nongaseous substances that can be ingested. These include carbon monoxide (CO), cyanide (CN), and hydrogen sulfide (HS). Systemic asphyxiants, on the other hand, work by interfering with oxygen transport or with intracellular utilization of oxygen. 1 Examples include carbon dioxide, nitrogen, fuels like propane and methane, and noble gases like argon. Simple asphyxiants include any gas that dilutes oxygen in ambient air and decreases the fraction of inspired oxygen (FiO2) below 21%. They are separated into two categories, simple and systemic asphyxiants, based on their mechanism of action (Figure 1). Although asphyxiant exposure is a relatively uncommon phenomenon, it is important to keep in mind when evaluating an altered patient or a patient who has been found down, particularly in industrial or fire-related incidents.Īsphyxiants are dangerous substances that deprive the body of oxygen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
